Story Highlight
– Many UK multivitamins fall short of Vitamin D recommendations.
– Labels misleadingly claim ‘100% of daily intake.’
– Current NRV for Vitamin D is outdated and insufficient.
– Confusion arises from varying serving sizes in products.
– Some brands plan to increase Vitamin D content soon.
Full Story
Concerns have emerged regarding the adequacy of Vitamin D levels found in various multivitamins, potentially misleading consumers about their nutritional intake, according to health experts. An investigation by a leading publication has revealed that many supplements available across the UK contain levels significantly lower than those recommended for adults, despite packaging claims of providing ‘100 per cent’ of daily needs.
Guidance from the National Health Service (NHS) encourages individuals aged four and above to consider a daily intake of 10 micrograms (mcg), particularly during the autumn and winter months when sunlight exposure, which aids in the body’s natural Vitamin D production, is reduced. Vitamin D, often referred to as the ‘sunshine vitamin’, is crucial for a variety of bodily functions including the regulation of calcium and phosphate, essential for maintaining healthy bones, teeth, and muscles. It can be sourced not only from sunlight but also from dietary sources such as oily fish, eggs, and cereals that have been fortified.
Despite these recommendations, the labels on many supplements still reference an outdated European nutrient reference value (NRV) of 5 mcg, established years ago and now deemed insufficient by UK standards. This discrepancy allows manufacturers to state that their products meet ‘100 per cent’ of daily Vitamin D requirements, while in reality, they deliver only half of what is now considered necessary by the government.
Dietitian Dr Carrie Ruxton, representing the Health & Food Supplements Information Service (HSIS), highlights the issues caused by this outdated regulation. “There is an unusual conflict in labelling laws, as the NRV is based on older European guidelines that do not align with current recommendations in the UK,” she explained. Consumers could mistakenly believe they are fulfilling their Vitamin D needs after purchasing a supplement that claims to meet their daily requirements. “In truth, 5 mcg falls short of what the UK Government advises. The NRV for Vitamin D is in need of revision and should be doubled,” she continued.
The analysis indicated that many mainstream multivitamin brands sold in retail outlets utilise the outdated NRV for their packaging. Particular concern surrounds gummy supplements, which tend to contain lower Vitamin D levels. Often, the number of capsules or gummies required to reach the stated dosage varies by brand, further complicating consumer understanding.
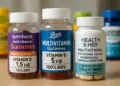
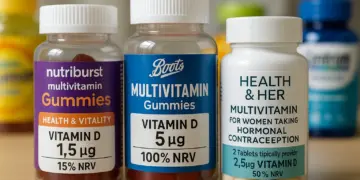
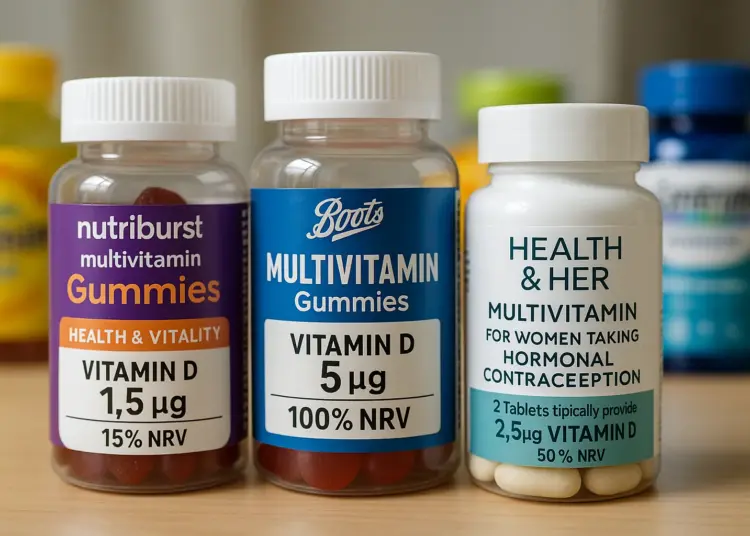
Notably, one product analysed, Nutriburst Health and Vitality Vitamin Gummies, provides a scant 1.5 mcg of Vitamin D per two gummies, which is only 15 per cent of the NHS’s recommended intake. Comparatively, Boots Multivitamin Gummies list 5 mcg per serving, while certain offerings from Health & Her include only 2.5 mcg. Confusingly, the packaging for Boots’ gummies states each gummy contains 5 mcg of Vitamin D3, representing ‘100 per cent’ of the NRV. For Health & Her’s products, two tablets yield 2.5 mcg, a mere 50 per cent of the NRV.
Variability in dosage can lead to misunderstandings surrounding Vitamin D intake, particularly among those who consume popular multivitamins which, according to the findings, often provide insufficient amounts of the vitamin contrary to what might be assumed from their labels.
Highlighting the other end of the spectrum, the investigation also identified a single multivitamin that delivered 45 mcg of Vitamin D per serving, exceeding the daily guidance yet remaining below the established safe upper limit for consumption. Consumers should also be aware that some supplements contain Vitamin D2, known as ergocalciferol, rather than Vitamin D3 (cholecalciferol), which research shows to be more effective in increasing Vitamin D levels in the bloodstream.
While sufficient dosages of Vitamin D are necessary, excessive intake can be harmful, leading to symptoms such as elevated calcium levels and potential kidney problems. However, these issues are generally linked to high-dose supplements and not from natural dietary sources or sun exposure.
Under the Food Supplements (England) Regulations of 2003, all food supplements must be labelled as such and contain vital information, including ingredients, recommended intake, and safety warnings. The clear stipulation that these products should not be a substitute for a balanced diet is also mandated on labelling.
In light of recent developments, the regulatory landscape for food supplements has undergone changes, especially following Brexit, when oversight transitioned to UK authorities. Nevertheless, there remains no universal upper limit set for all vitamins and minerals across the UK, with safety evaluations determined through scientific guidance instead.
In response to the investigation, representatives from various brands provided insights. Tarun Kapoor, Chief Operating Officer at Nutriburst, clarified that their Health & Vitality Multivitamin Gummies do not assert to be a complete source of Vitamin D. He reiterated their commitment to clear labelling and product improvements, noting the forthcoming releases aimed at higher Vitamin D levels.
Dr Robin Andrews from Health & Her explained the formulation of their products is based on evidence concerning nutrient metabolism in women using hormonal contraception or hormone replacement therapy, resulting in a conservative approach to Vitamin D inclusion.
Mondelez International, which oversees Bassetts Multivitamins, stated compliance with regulatory requirements and mentioned that they are increasing Vitamin D levels across their adult product range.
Overall, these revelations underscore the importance of consumers understanding the actual nutrient content in supplements and how age-old guidelines can lead to misunderstandings about dietary needs, particularly concerning critical vitamins like Vitamin D.
Our Thoughts
To avert the confusion regarding vitamin D intake from multivitamins, clearer regulatory standards aligned with current UK health guidance are essential. The discrepancy in labelling laws, where products containing 5mcg of Vitamin D can misleadingly claim to fulfill ‘100% of daily requirements,’ needs reevaluation under the Food Supplements (England) Regulations 2003. This legislation mandates accurate and non-misleading information on labels, which brands are currently failing to meet with outdated nutrient reference values.
A key safety lesson is the need for consistent updating of nutritional guidelines within product labelling to reflect current health recommendations. Additionally, proper consumer education about vitamin D intake, including the variation in types and dosages of supplements, is crucial for informed choices.
Companies should ensure that their products meet the latest recommendations and provide explicit, accurate dosing information. Regular audits and the enforcement of stringent penalties for misleading claims could help in maintaining consumer safety. Ensuring compliance with the UK Nutrition and Health Claims Regulation is vital to prevent similar incidents in the future.