Story Highlight
– Napralief painkiller recalled for missing dosage information.
– Affected batches include numbers B51496, B51497, B51102.
– Missing instructions may lead to unsafe usage.
– Patients advised to report adverse reactions promptly.
– MHRA mandates return of remaining stock to suppliers.
Full Story
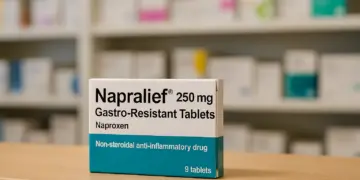
The United Kingdom has seen the withdrawal of a popular pain relief medication due to significant omissions in its dosage information on packaging. In a move driven by safety concerns, the Medicines and Healthcare products Regulatory Agency (MHRA) has prompted the recall of specific batches of Napralief 250mg Gastro-Resistant Tablets, produced by Omega Pharma Limited.
The affected product, a non-steroidal anti-inflammatory drug (NSAID), contains the active ingredient naproxen, commonly used to alleviate pain from various conditions, including muscle aches, joint pain, inflammation from sports injuries, lower back discomfort, neck stiffness, and menstrual pain.
The recall specifically concerns batches numbered B51496, B51497, and B51102, all of which are now flagged for immediate removal from store shelves and healthcare providers’ inventories.
Patients taking Napralief are urged to seek advice from healthcare professionals if they encounter any side effects or have queries regarding their treatment. The MHRA has highlighted that the impacted batches lack crucial information regarding the recommended maximum daily dosage of three tablets. This warning is vital as it aims to prevent the risk of overconsumption, which can lead to adverse effects.
Among the omissions in the patient information leaflet are necessary dosage instructions for initial and subsequent dosing. According to standard guidance, users should take two tablets on the first day and then one tablet every six to eight hours afterward, if needed. The protocol for the second and third days similarly advises taking one tablet within the same time frame, and it is important to note that Napralief should not be used for longer than three consecutive days without medical consultation.
Furthermore, the leaflet failed to provide essential cautions regarding several serious health concerns. This includes the recommendation for patients to seek an eye examination should they experience any visual disturbances, highlighting the risks of potential allergic reactions that can occur even in individuals who have previously not shown any sensitivity to such medications.
Additional warnings, such as informing a healthcare provider if blood or urine tests are forthcoming, which requires stopping the medication 48 hours before testing, were also missing from the instructions. Moreover, critical information related to heart conditions, various autoimmune diseases, and potential severe skin reactions was not adequately presented, potentially leaving patients at risk due to insufficient understanding of the medication’s use.
Dr. Alison Cave, the Chief Safety Officer at the MHRA, addressed the situation, affirming the safety of Napralief 250mg when used according to the correct dosage guidelines. She stated, “Napralief 250mg is considered safe when used in line with the correct dosage instructions. Although small unintentional dosing mistakes are usually not harmful, complete and accurate safety information is essential to help ensure patients use their medicine correctly.”
In her message to the public, Dr. Cave reiterated the correct usage of the medication: “Patients can continue to use the medicine safely in line with the correct safety and dosage instructions. Patients should take two tablets on the first day, followed by one tablet 6-8 hours later. For the second and third day of treatment, if needed, one tablet should be taken every 6–8 hours. Napralief should not be taken for more than three days.”
In light of these developments, the MHRA has encouraged healthcare professionals to cease the distribution of the affected batches immediately and ensure that any remaining stock is returned to suppliers. This precautionary approach is intended to uphold patient safety and ensure that vital health information is conveyed comprehensively.
Patients who experience adverse reactions while taking Napralief are advised to report these incidents through the MHRA’s Yellow Card scheme, contributing to ongoing safety surveillance and allowing the agency to monitor the situation closely.
As the situation unfolds, it serves as a stark reminder of the importance of accurate medication guidelines and the critical need for rigorous checks within pharmaceutical packaging to guarantee patient safety. The implications of missing safety information are significant, warranting swift action from healthcare authorities to protect public health effectively.
In conclusion, this recall highlights the vital role of clear communication in healthcare, particularly regarding medication safety. Patients are urged to stay informed, seek guidance when uncertain, and always adhere to the prescribed guidelines for their medications.
Our Thoughts
The recall of Napralief 250mg Gastro-Resistant Tablets highlights significant regulatory compliance failures under UK health and safety legislation. Key dosage instructions and critical safety information were missing from the product packaging, which is a breach of the Consumer Protection Act 1987, as it did not ensure that the product was safe for consumer use.
To prevent this incident, Omega Pharma Limited should have implemented a more rigorous quality control and compliance review process before the product’s release. Enhanced training for employees on the importance of accurate packaging in adherence to the Medicines and Healthcare products Regulatory Agency (MHRA) guidelines could further mitigate risks.
Additionally, regular audits and checks should be established to ensure that all safety instructions, including cautions for specific medical conditions, are clearly communicated to consumers. This incident serves as a vital reminder that companies must maintain thorough documentation and clear communication related to drug safety information, thereby prioritizing patient safety and compliance with health regulations.