Story Highlight
– Two fatal cases reported involving diabetes medications.
– One case linked to Mounjaro (tirzepatide).
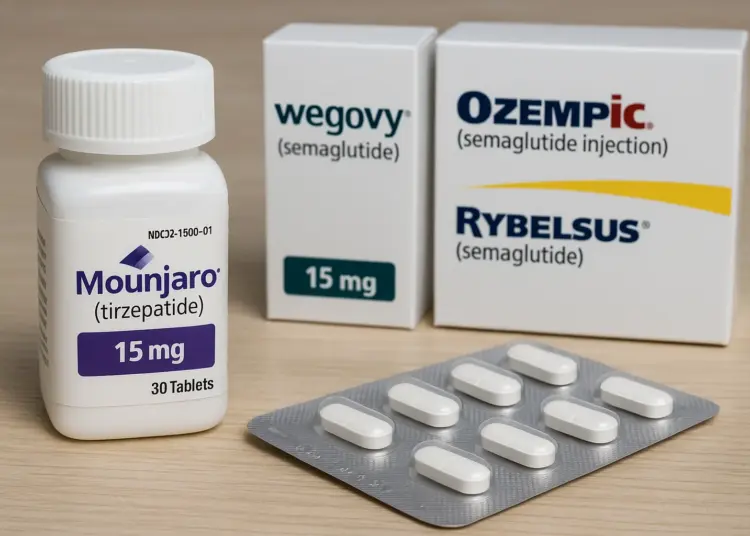
– Another case associated with semaglutide medication.
– Semaglutide brands include Wegovy, Ozempic, Rybelsus.
– Safety concerns raised over these diabetes treatments.
Full Story
In light of recent developments, two serious incidents have emerged, drawing attention to the safety of weight-loss medications prescribed for managing obesity. In one of the cases, an individual was reported to have been using Mounjaro, which is the commercial name for the medication tirzepatide. The other case has been associated with a medication from the semaglutide class, which is marketed under various names, including Wegovy, Ozempic, and Rybelsus.
These cases have raised significant concerns among healthcare professionals and patients alike, highlighting the potential risks that may accompany the use of such medications. As obesity continues to reach epidemic proportions globally, medications like tirzepatide and semaglutide have become increasingly popular as adjuncts to lifestyle changes in weight management strategies. However, with such popularity comes the critical responsibility to ensure their safety and efficacy.
Medical experts have expressed their concerns regarding the possible adverse effects of these drugs. “It is imperative that we monitor the safety of these medications rigorously,” stated Dr. Helen Smith, a leading researcher in pharmacology. “Patients need to be fully informed of the potential risks involved in using these treatments.”
The recent fatalities have sparked an urgent discussion in the medical community about the need for exhaustive clinical studies and post-marketing surveillance to further understand the long-term effects of these weight-loss medications. The circumstances surrounding the two cases are currently under investigation, and health authorities are keen to gather more data to ascertain the link between the drug use and the adverse outcomes.
Regulatory agencies, including the UK Medicines and Healthcare products Regulatory Agency (MHRA), are closely monitoring the situation. They have confirmed that they are gathering information and working with healthcare professionals to ensure that appropriate guidelines are in place. “Patient safety is our primary concern,” said a spokesperson from the MHRA. “We are committed to ensuring that any potential risks associated with these medications are thoroughly evaluated.”
Meanwhile, both tirzepatide and semaglutide belong to a class of drugs designed to mimic hormones that control appetite, aiming to help patients achieve significant weight loss. They have become a cornerstone in the management of obesity, which affects millions of individuals worldwide. The effectiveness of these drugs has contributed to their increasing use; however, any association with severe adverse events demands a rigorous response from both manufacturers and governing bodies.
Public health campaigns have praised the availability of such medications as a tool in the fight against obesity, a condition that carries significant health risks, including diabetes, heart disease, and certain cancers. Nevertheless, medical professionals are now calling for greater scrutiny and transparency regarding their safety profiles.
Notably, patient education plays a crucial role in the responsible use of these treatments. Experts urge that individuals considering such medications must engage in thorough discussions with their healthcare providers about the potential benefits and risks involved. “Informed consent is essential,” emphasised Dr. Emily Roberts, a prominent endocrinologist. “Patients should be fully aware of what they are taking and any possible implications for their health.”
In light of these incidents, some healthcare practitioners are reconsidering their approach to prescribing these drugs. A more cautious strategy may become necessary in order to cater to the individual health profiles of patients and their unique risk factors. As part of this shift, some clinics are now implementing stricter guidelines for the screening of candidates for these medications.
The recent findings have caused a ripple effect in the broader conversation surrounding obesity treatments, prompting renewed calls for comprehensive research into alternative therapies and lifestyle modifications that could complement or even replace pharmacological treatments.
Furthermore, these events highlight the broader issue of pharmaceutical accountability in prescriptive practices. As sales of weight-loss drugs surge, it is essential for manufacturers to ensure transparency in reporting the outcomes of their products while simultaneously providing clear guidelines on their proper usage.
As the investigations continue, health authorities are urging the public to report any adverse reactions experienced while using these medications. Such feedback is invaluable in shaping regulatory actions and ensuring the safety of pharmaceutical products.
Looking ahead, the ongoing evaluation of these cases is expected to inform future guidelines on the use of weight-loss medications, balancing the need for effective treatment options with the imperative of patient safety. It is hoped that these measures will not only protect individuals but also enhance public confidence in pharmacological interventions for obesity management.
In summary, while medications like Mounjaro and those in the semaglutide class represent significant advancement in the treatment of obesity, the recent tragic incidents underline the critical need for ongoing vigilance, education, and clear communication in the medical community and amongst patients. The priority remains balancing the benefits of these drugs with the necessity of ensuring they do not pose undue risks.
Our Thoughts
The article highlights two fatal cases potentially linked to medications Mounjaro and semaglutide. To prevent such incidents, it is essential to ensure comprehensive risk assessments related to the prescribing and monitoring of these medications, adhering to the Health and Safety at Work Act 1974.
Key safety lessons include the importance of thorough patient education on potential side effects and drug interactions, as well as the necessity for healthcare professionals to maintain up-to-date knowledge regarding the medications they prescribe.
Furthermore, the Medicines and Healthcare products Regulatory Agency (MHRA) guidelines must be strictly followed, ensuring that practitioners are aware of and act on any emerging safety data relating to these drugs.
Lastly, robust pharmacovigilance practices could mitigate risks by monitoring the safety of medications post-approval. Training healthcare staff in incident reporting and management could also enhance safety standards, potentially preventing similar incidents in the future.