Story Highlight
– Batch of Sertraline recalled after packaging mix-up.
– Incorrectly packaged Citalopram found in Sertraline cartons.
– Pharmaceutical company Amarox issued precautionary recall.
– MHRA advises patients to check medication packaging.
– Contact pharmacy if Citalopram is found in cartons.
Full Story
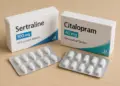
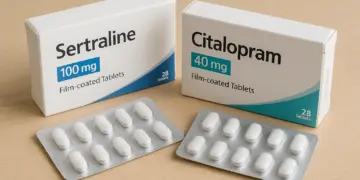
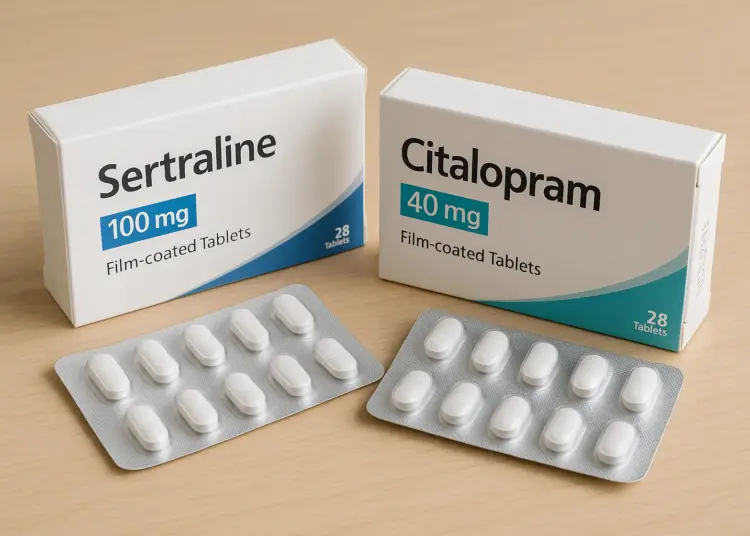
Health authorities have taken decisive action to recall a particular batch of a widely prescribed antidepressant, following a troubling incident involving a mix-up in medication. The issue surfaced when a patient reported the unexpected presence of a different antidepressant within a sealed package of Sertraline 100mg film-coated tablets.
This alarming discovery involved the presence of Citalopram 40mg film-coated tablets nestled inside a carton intended solely for Sertraline. The incident has raised serious concerns regarding patient safety, as it highlights the possibility of a packaging error that could affect others who might have received medication from the same batch.
In light of this situation, Amarox, the UK-based pharmaceutical company responsible for the production of Sertraline and Citalopram, has announced a “precautionary recall” of the specific batch concerned. This decision has been supported by the Medicines and Healthcare products Regulatory Agency (MHRA), which stated that the recall is being conducted as a safety measure while further investigations are undertaken to determine the cause of the mix-up.
The MHRA has clarified that both Sertraline and Citalopram are produced by the same manufacturer at identical production facilities. The agency’s investigation suggests that the error likely occurred during the secondary packaging phase, which means the production of the tablets themselves was not at fault. Instead, the mix-up transpired after the tablets had already been manufactured and packaged.
Patients in possession of the affected batch, which has been assigned the batch number V2500425, are advised to carefully inspect their medication to ensure it is correct. Health officials have urged individuals to remain cautious and vigilant. Anyone who suspects they might have inadvertently ingested Citalopram 40mg tablets instead of their prescribed Sertraline is advised to seek immediate medical guidance, particularly if they are experiencing unusual side effects.
Pharmacists across the country have been informed by the MHRA to halt the distribution of the implicated batch without delay. They have also been instructed to reach out to patients who may have received the incorrect medication and provide appropriate instructions.
Dr. Alison Crave, the chief safety officer at the MHRA, emphasised the importance of patients verifying the contents of their medication packaging. She stated, “If you have been prescribed Sertraline 100mg tablets and have received batch number V2500425, please check the carton contains the right medication. You can find the batch number and expiry date printed on the side of the outer packaging. If the blister strips inside the carton are labelled Citalopram 40mg, please contact your pharmacy as soon as possible. If they are labelled Sertraline 100mg, no further action is needed.”
In her remarks, Dr. Crave highlighted the potential ramifications of mistakenly consuming Citalopram, either instead of or alongside Sertraline, which could lead to heightened serotonergic side effects. These side effects may manifest as symptoms such as nausea, headaches, changes in sleep patterns, and mild anxiety. While these symptoms are typically not severe for most individuals, Dr. Crave underscored the importance of seeking medical advice if patients are at all concerned about their health.
The incident serves as a stark reminder of the critical role that proper packaging and quality control play in the pharmaceutical industry. Sertraline is one of the most commonly prescribed antidepressants in the UK; in 2019 alone, general practitioners in England issued over 16.7 million prescriptions for this medication. The high prescription rate underscores the reliance many patients have on these medications for their mental health management.
Health professionals and industry experts are likely to scrutinise this incident closely to safeguard against future occurrences. The recall has prompted discussions about the potential need for enhanced safety measures in the packaging processes for medications, particularly those that have such a significant impact on patient health.
As investigations proceed, the MHRA continues to monitor the situation closely to ensure that patient safety remains the top priority. In the meantime, both the agency and healthcare providers are focused on ensuring that affected patients receive the necessary support and information to manage any potential risks associated with the mix-up.
For the general public, the incident serves as a poignant reminder of the importance of being aware of the medications they are taking and the necessity of adhering to best practices when it comes to prescription medications. Patients are encouraged to remain proactive in their healthcare, including asking questions and seeking clarification whenever they have concerns about their prescribed treatments.
In summary, the precautionary recall of the Sertraline batch is a significant development that underscores the healthcare system’s commitment to patient safety. As the investigation unfolds, it is critical for individuals to remain vigilant and informed about their medications, helping to prevent any negative health outcomes linked to such packaging errors.
Our Thoughts
The incident described highlights critical failures in the pharmaceutical packaging process, specifically during secondary packaging, potentially breaching the Medicines and Healthcare products Regulatory Agency (MHRA) guidelines and the Health and Safety at Work Act 1974, which emphasizes ensuring the safety of products provided to consumers.
To prevent such incidents, pharmaceutical manufacturers should implement stricter quality control measures and routine audits of their packaging processes to ensure that only the correct medications are placed in respective cartons. Additionally, employing advanced packaging technology, such as automated verification systems, could significantly reduce the risk of human error.
Training staff involved in the packaging process on the importance of adherence to material handling protocols and promoting a culture of safety can also reduce errors. Regular reviews of processes and immediate incident reporting mechanisms can help identify and rectify potential failures before they impact patients.
Further, clearer communication to patients about the recall process and confirming their medications could enhance safety and trust in pharmaceutical practices. These steps are imperative to avoid similar future incidents and maintain compliance with UK health regulations.